POSTED ON
Jul 29, 2025

Today, we’re proud to announce TREAT-2, our second fine-tuned model in the TREAT (Targeted Reward Evaluation and Therapeutics) initiative, this time focused on epigenetic targets.
Where TREAT-1 mapped neurotransmitter transporters, TREAT-2 zeroes in on enzymes that govern gene expression itself, Histone deacetylases, or HDACs, offering a lens into the switches and levers that tune human biology at its most fundamental layer.
🔬 Why it matters: In diseases ranging from Alzheimer’s to autoimmune disorders to glioblastoma, gene expression is dysregulated—turned on in the wrong places or silenced when it should be active. TREAT-2 helps identify molecules that can selectively bind and modulate these gene expression controllers, bringing the dream of epigenetic precision medicine one step closer.
🔥 Model highlights:
TREAT-2: fine-tuned from PSICHIC, our base model, using a highly curated dataset of histone deacetylase (HDAC) interactions.
Incorporates both human and rat data to enhance cross-species translation—vital for preclinical pipeline progression.
Achieved a 23% drop in Mean Average Prediction Error and a 45% reduction in variance across validation folds signaling a leap in the model’s ability to generalize across novel chemical scaffolds. This means TREAT-2 doesn’t just memorize known interactions, it can predict new ones, dramatically increasing our chances of finding real therapeutic hits.
Introduction
What Are HDACs and Why Do They Matter?
HDACs are enzymes found throughout our bodies, with 18 different types in humans [1]. They work by removing acetyl groups from proteins called histones, which act like spools that DNA wraps around. When HDACs remove these acetyl groups, DNA becomes more tightly wound and harder to read. This process helps control which genes are active in our cells at any given time [2].
At Metanova Labs, within the TREAT (Targeted Reward Evaluation and Therapeutics) initiative, our interest lies in exploring the function of HDACs in the brain. This research aims not only to facilitate the development of novel treatments for neurological and psychiatric disorders but also to potentially enhance the quality of life and extend healthspans.
In a healthy brain, HDACs work in balance with other enzymes to ensure the right genes are turned on or off at the right times. This balance is essential for learning, memory formation, and mood regulation [3]. However, research has shown that this balance is often disrupted in several psychiatric and neurological conditions. For example, in depression, certain HDACs may be overactive, shutting down genes that normally help brain cells communicate and adapt to stress [4].
The Promise of HDAC Inhibitors
Scientists have developed drugs called HDAC inhibitors that can block these enzymes, potentially reopening silenced genes and restoring normal brain function. One particularly exciting aspect is that HDAC inhibitors don't randomly activate genes. Instead, they create a "primed" state where genes become more responsive to normal brain signals, like those generated during learning or therapy, which makes them potentially powerful tools for enhancing other treatments [5].
Research has revealed that HDAC dysfunction contributes to many neurological and psychiatric disorders beyond depression:
Neurodegenerative Diseases
In Huntington's disease, HDAC3 inhibitors not only prevent memory impairment but may actually slow disease progression by reducing pathological gene expansions [9]
For ALS, HDAC6 inhibitors help restore the cellular "highways" that transport essential supplies within motor neurons, potentially preserving nerve-muscle connections [16]
In multiple sclerosis, HDAC inhibitors serve double duty - calming destructive inflammation while promoting repair of damaged myelin insulation around nerves [19]
Addiction and Memory
HDAC3 acts as a "molecular brake" on forgetting drug-associated memories - removing this brake helps the brain "unlearn" addiction. When combined with behavioral therapy, HDAC inhibitors make the brain more receptive to forming new, healthier associations. This mechanism could revolutionize addiction treatment by enhancing the effectiveness of counseling and rehabilitation programs. [17]
Cognitive Enhancement and Aging
HDAC inhibitors don't directly increase intelligence but optimize the brain's ability to form and strengthen memories during learning [5]
In aging brains, these drugs can restore youthful patterns of gene expression, potentially maintaining cognitive sharpness [18]
The enhancement is specific to learning actively - the drugs amplify the benefits of training and education rather than creating random changes [5]
Developmental and Social Disorders
In autism models, selective HDAC inhibitors improve social behaviors and reduce repetitive actions by normalizing synaptic gene expression [20]
Timing is critical - while prenatal HDAC inhibition increases autism risk, postnatal treatment shows therapeutic promise [20]
These findings suggest potential for improving social cognition and behavioral flexibility in neurodevelopmental conditions
This broad therapeutic potential reflects HDACs' fundamental role in regulating neural gene expression across multiple brain systems and throughout the lifespan.
The Challenge of Translation
Despite these promising laboratory and animal model findings, bringing HDAC inhibitors to patients has its challenges. The first generation of these drugs was originally developed for cancer treatment and came with significant side effects. Up to 98% of patients experienced drops in blood platelet counts, along with severe fatigue and digestive problems [7]. These side effects might be acceptable for cancer patients, but they're far too severe for treating chronic brain conditions.
Another major hurdle is getting these drugs into the brain. The blood-brain barrier, which protects our brains from harmful substances, also keeps out many medications. Early HDAC inhibitors had poor brain penetration and were quickly eliminated from the body, requiring frequent dosing that made long-term treatment impractical [8].
New Approaches and Innovations
Some of the innovative approaches that aim to overcome these challenges include:
Selective Targeting: New drugs target specific HDAC types rather than blocking all of them. For instance, HDAC3-selective inhibitors focus on memory enhancement, while HDAC6 inhibitors target nerve cell protection. This selectivity reduces side effects while maintaining benefits [9,10].
Better Delivery Systems: Researchers are developing special formulations that help drugs cross the blood-brain barrier. These include nanoparticle carriers and "pro-drugs" that only become active once they reach the brain [12].
Combination Therapies: Rather than using HDAC inhibitors alone, scientists are exploring how they might enhance existing treatments. For example, combining them with psychotherapy for PTSD or with antidepressants for major depression [13].
The screening of ultra large libraries of chemical compounds performed by miners at NOVA has the potential to uncover drug candidates that act more selectively over the desired subtype of HDAC and present more more desirable ADMET (Absorption, Distribution, Metabolism, Excretion and Toxicity) profiles.
Recent Clinical Progress
While we're still in the early stages, there have been encouraging developments. In 2024, the FDA approved givinostat (Duvyzat) for Duchenne muscular dystrophy, marking the first approval of an HDAC inhibitor for a non-cancer condition [14]. This milestone proves that with careful drug design, HDAC inhibitors can be safe enough for long-term use.
Early clinical trials in psychiatric conditions are also underway. Some studies combine HDAC inhibitors with existing treatments, hoping to boost their effectiveness [15]. Others explore whether these drugs might help "lock in" the benefits of psychotherapy by enhancing the brain's ability to form new, healthier neural pathways.
TREAT-2: Advanced binding predictions for HDACs
In continuation to the work performed on TREAT-1 and building upon the knowledge gained during its development, we have continued to build upon the PSICHIC (PhySIchoCHemICal graph neural network) architecture.
PSICHIC is a state-of-the-art model that receives sequence-only data and matches or even surpasses the performance of other, much more computationally expensive methods in predicting how different chemical compounds will interact with protein targets of interest. Accurately modeling the likelihood of the interaction between small molecules and proteins is central to modern drug discovery, since these interactions dictate pharmacological efficacy and selectivity, as well as off-target side effects. Reliable in-silico models also help prioritize the most promising compounds for costly experimental validation, potentially compressing discovery timelines and use of resources in the search for next generation therapeutics.
For TREAT-2, similarly to TREAT-1, we have performed a domain-specific fine-tuning with data augmentation (DS-FT + A) on the original PSICHIC pretrained model for 8 subtypes of human and mice HDACs. The dataset used for the fine-tuning process contained ~6.3k data points that were already present in the pretraining dataset, to which we added around another 6k labeled data points carefully curated from the literature, patents and other scientific publications.
The dataset was partitioned into five Bemis–Murcko scaffold folds, ensuring every chemical scaffold appeared in only one partition. This is done to encourage model generalization to novel chemical structures instead of learning the characteristics of a few specific chemical moieties. For each fold, compounds in the held-out set served as external validation while the remaining four folds were used for fine-tuning the model.
A series of experiments was conducted to assess the ideal training parameters for these specific targets. The hyperparameters analyzed included the depth of fine-tuning (i.e., the number of tunable layers), as well as learning rate and number of iterations to avoid overfitting. The baseline model and all fine-tuned variants were assessed on the five external validation folds. Here, we present the model that showed the best performance in the held-out sets in comparison to the baseline.
First, we will analyze the coefficient of determination (R2) between true and predicted values. This value ranges from 0 to 1, with 0 meaning no correlation between the two sets of values and 1 meaning perfect correlation. The baseline model has a median R2 of 0, meaning that it had basically no predictive power for this family of targets. Fine tuning the model with data augmentation raised this value to 0.32, a significant improvement:
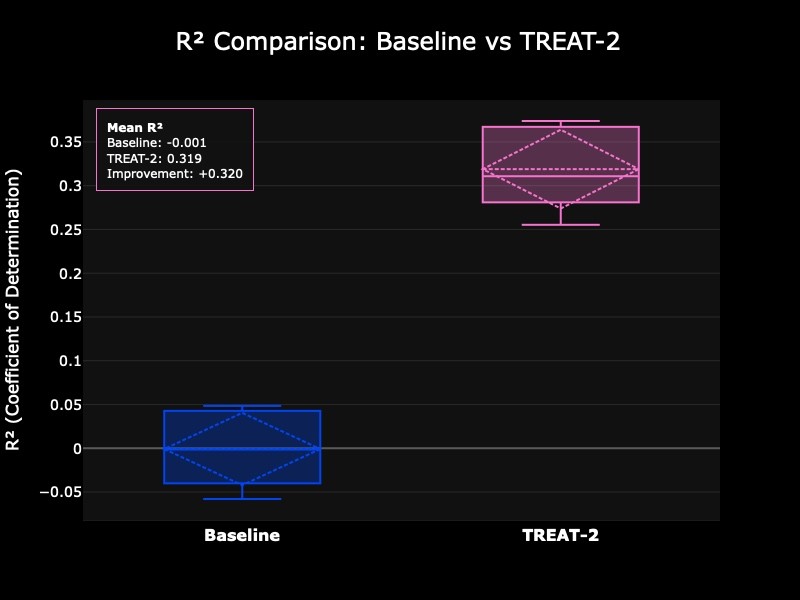
Figure 1: R2 comparison between baseline and fine-tuned models across five folds.
Next, we will analyze the Mean Absolute Percentage Error (MAPE), which measures the average error (in percentage, that is, agnostic to scale) between true and predicted values. This value ranges from 0% to 100%, and decreasing it means improving the model, since the error is smaller. For TREAT-2 not only did we obtain a 23% reduction in MAPE, but also variance between folds is 45% smaller, indicating improved generalizability on different chemical scaffolds by a large margin.
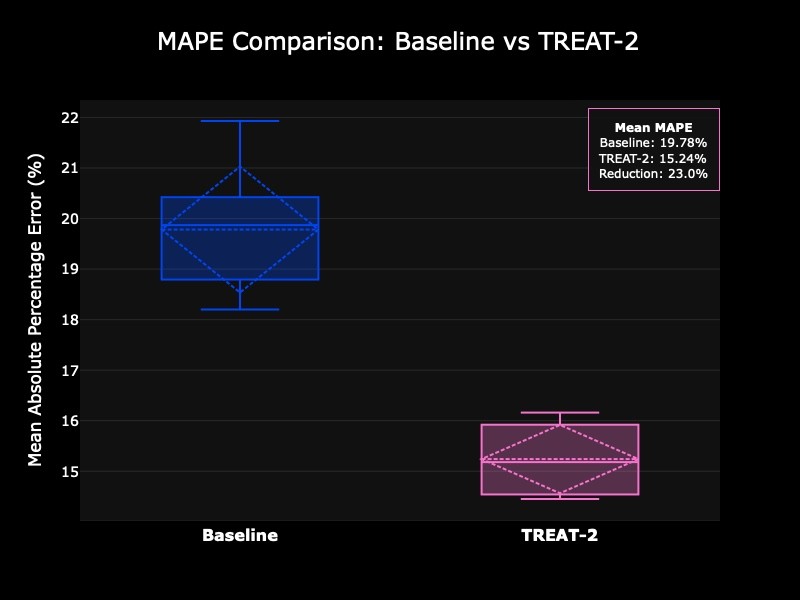
Figure 2: Mean Average Percentage Error comparison between baseline and fine-tuned models across five folds.
This means the model is no longer limited to patterns it has seen before; it can now more reliably extrapolate to structurally novel molecules. These gains represent a major leap forward in our ability to identify previously inaccessible chemical space and accelerating the discovery of new therapeutic compounds for high-impact epigenetic targets.
Conclusion
HDACs are a family of targets with huge, unexplored potential for the treatment of several neuropsychiatric disorders, as well as for the improvement in quality of life for people who can benefit from behavioral modulation, attenuation of the normal cognitive decline associated with aging, as well as enhanced learning abilities. Being able to reliably model how small molecules will interact with this class of proteins may facilitate the development of next generation therapeutics and TREAT-2 shows significant improvement over the general version of the state-of-the-art model PSICHIC for this family of targets.
By fine-tuning the baseline model with an augmented dataset of carefully curated, labeled samples, we were able to unlock a family-specific signal previously invisible to the general-purpose model, significantly increasing its predictive power, reducing Mean Absolute Percentage Error in 23% and reducing Inter-fold Variation in 45%. This means more accurate and consistent predictions that are better suited to be used in the prioritization of chemical compounds for drug development.
TREAT-2 has been deployed in NOVA where it will be used in a highly adversarial ecosystem to screen an ultra large molecular library in the search for the compounds with the highest likelihood of having a desired effect over specific HDACs.
—
References
[1] Curcio A, et al. The Histone Deacetylase Family: Structural Features and Application of Combined Computational Methods. Pharmaceuticals. 2024;17(5):620.
[2] Yang W, Seto E. A short guide to histone deacetylases including recent progress on class II enzymes. Experimental & Molecular Medicine. 2021;53:1-12.
[3] Burns AM, Gräff J. Cognitive epigenetic priming: leveraging histone acetylation for memory amelioration. Current Opinion in Neurobiology. 2021;67:75-84.
[4] Park HS, et al. Epigenetic Targeting of Histone Deacetylases in Diagnostics and Treatment of Depression. International Journal of Molecular Sciences. 2021;22(10):5398.
[5] Burns AM. Epigenetic memory aids: Synaptic and molecular effects of HDAC inhibition that support memory formation. EPFL Thesis. 2022.
[6] Chen S, Li S. Epigenetic Regulation in Alzheimer's Disease–Development, Challenges, and Future Perspectives. Scientific Archives. 2025.
[7] Subramanian S, et al. Clinical toxicities of histone deacetylase inhibitors. Pharmaceuticals. 2010;3(9):2751-2767.
[8] Wey HY, et al. Image-guided synthesis reveals potent blood-brain barrier permeable histone deacetylase inhibitors. ACS Chemical Neuroscience. 2014;5(8):689-698.
[9] Kirkham-McCarthy L, et al. A selective inhibitor of HDAC3 prevents cognitive deficits and suppresses striatal CAG repeat expansions in Huntington's disease mice. Scientific Reports. 2017;7:6082.
[10] Wexler M. New HDAC6 inhibitor shows myelination potential in CMT1A models. Charcot-Marie-Tooth News. September 22, 2020.
[11] Xiao Y, et al. HDAC3 and HDAC8 PROTAC dual degrader reveals roles of histone acetylation in gene regulation. Cell Chemical Biology. 2023;30(11):1421-1435.
[12] Gladkova MG, et al. Epidrugs in the Therapy of Central Nervous System Disorders: A Way to Drive on? Cells. 2023;12(11):1464.
[13] Bonomi RE, et al. A Role for Histone Deacetylases in the Biology and Treatment of Post-Traumatic Stress Disorder: What Do We Know and Where Do We Go from Here? Complex Psychiatry. 2022;8:13-27.
[14] FDA News Release. FDA Approves Nonsteroidal Treatment for Duchenne Muscular Dystrophy. March 21, 2024.
[15] Smith RC. Sodium Butyrate For Improving Cognitive Function In Schizophrenia. ClinicalTrials.gov Identifier: NCT03176472. 2016.
[16] Taes I, et al. Hdac6 deletion delays disease progression in the SOD1G93A mouse model of ALS. Human Molecular Genetics. 2013;22(9):1783-1790.
[17] Malvaez M, et al. HDAC3-selective inhibitor enhances extinction of cocaine-seeking behavior in a persistent manner. Proceedings of the National Academy of Sciences. 2013;110(7):2647-2652.
[18] Gräff J, et al. An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature. 2012;483(7388):222-226.
[19] Faraco G, et al. The therapeutic potential of HDAC inhibition in multiple sclerosis. Molecular Medicine. 2011;17(5-6):442-447.
[20] Qin L, et al. Social deficits in Shank3-deficient mouse models of autism are rescued by histone deacetylase (HDAC) inhibition. Nature Neuroscience. 2018;21(4):564-575.